Financial Conflict of Interest (FCOI) Policy
Updated September 2025 – (Applies to investigators submitting NIH/NSF research applications on or after August 24, 2012 as well as to PIs on active NIH and NSF awards)
Introduction. In accordance with Public Health Service (PHS) regulation 42 CFR Part 50 Subpart F (grants and cooperative agreements) and 45 CFR Part 94 (contracts), and consistent the with the provisions of the National Science Foundation (NSF) Proposal & Award Policies & Procedures Guide, Part II, Award & Administration Guide (AAG) Chapter IV.A, Kalamazoo College has developed the following policy on financial conflict of interest (FCOI) for investigators applying for or holding grants from the National Institutes of Health (NIH) or the National Science Foundation (NSF). The PHS and NSF requirements were “designed to promote objectivity in PHS- [and NSF-] funded research by establishing standards to ensure that the design, conduct, and reporting of research […] is not biased by any conflicting financial interests of an Investigator […]. The regulation places the responsibility for the identification and management of investigators’ FCOI with the institution, which oversees the Investigator’s activities.”
Scope. The following statement of policy applies to Kalamazoo College investigators applying to or holding grants from the NIH or the NSF.
The term investigator means the principal investigator, co-principal investigators/co-project directors, and any other person at the institution who is responsible for the design, conduct, or reporting of research activities funded or proposed for funding by the NIH or the NSF.
Fiduciary responsibility. Members of the Kalamazoo College community serve the public trust and thus have a clear obligation to conduct the business of the College in a manner consistent with those responsibilities. All decisions of the board, officers of the College, faculty, and staff are to be made solely on the basis of a desire to promote the best interests of the College and the public good.
In the spirit of the public trust, faculty or staff proposing to receive funds from the NIH or the NSF for research activities must disclose – before submitting a proposal – any substantive financial conflict of interest that may reasonably affect how the project is carried out. A possible conflict of interest does not preclude acceptance of NIH or NSF funding, but appropriate safeguards may need to be implemented in certain situations.
Institutional official. Kalamazoo College designates the Director of Faculty Grants as the institutional official who reviews financial disclosure statements from each investigator who is proposing or carrying out NIH- or NSF-funded research.
Process to inform investigators. The Director of Faculty Grants will direct the investigator to the FCOI policy, including the training requirement.
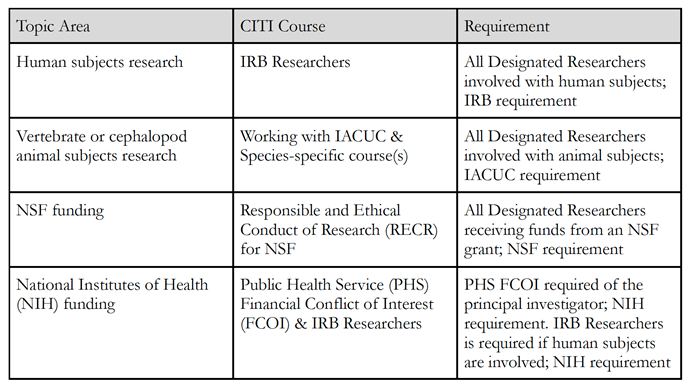
Training. Investigators must receive FCOI training before submitting an NIH/NSF application and repeat the training at least once every four years. Training must take place right away if a) an investigator
is new to Kalamazoo College; b) Kalamazoo College revises its FCOI policy in a way that affects requirements of investigators; or c) the investigator is not in compliance with the policy. Investigators can find the NIH FCOI training module at the following link: https://grants.nih.gov/grants/policy/coi/tutorial2018/story_html5.html
Upon successful completion of the training module, the investigator must print a copy of the certificate of
completion, keep one copy, and send another copy to the Director of Faculty Grants (the Institutional
Official).
Disclosure of Significant Financial Interest (SFI). Before submitting a grant proposal to the NIH or the NSF, the investigator must complete the College’s Financial Disclosure Form and turn it in to the Director of Faculty Grants. On the form, the investigator must report all significant financial interests (including those of his or her spouse or domestic partner and dependent children)
- that would reasonably appear to be affected by the research activities; or
- in entities whose financial interests would reasonably appear to be affected by such activities.
The proposal may not be submitted until the Financial Disclosure Form has been completed and turned in.
Significant Financial Interest Definition: SFI is defined by the regulation as:
- A financial interest consisting of one or more of the following interests of the investigator (and those of the investigator’s spouse or domestic partner and dependent children) that reasonably appears to be related to the investigator’s institutional responsibilities:
- With regard to any publicly traded entity, a significant financial interest exists if the value of any remuneration received from the entity in the twelve months preceding the disclosure and the value of any equity interest in the entity as of the date of disclosure, when aggregated, exceeds $5,000. Remuneration includes salary and any payment for services not otherwise identified as salary (e.g., consulting fees, honoraria, paid authorship); equity interest includes any stock, stock option, or other ownership interest, as determined through reference to public prices or other reasonable measures of fair market value;
- With regard to any non-publicly traded entity, a significant financial interest exists if the
value of any remuneration received from the entity in the twelve months preceding the
disclosure, when aggregated, exceeds $5,000, or when the investigator any equity interest
(e.g., stock, stock option, or other ownership interest); or - Intellectual property rights and interests (e.g., patents, copyrights), upon receipt of income related to such rights and interests.
- Investigators also must disclose the occurrence of any reimbursed or sponsored travel (i.e., that which is paid on behalf of the investigator and not reimbursed to the investigator so that the exact monetary value may not be readily available) related to their institutional responsibilities; provided, however, that this disclosure requirement does not apply to travel that is reimbursed or sponsored by a United States (US) federal, state, or local government agency, a US institution of higher education as defined at 20 U.S.C. 1001(a), a US academic teaching hospital, a US medical center, or a US research institute that is affiliated with an institution of higher education. Disclosure will include, at a minimum, the purpose of the trip, the identity of the sponsor/organizer, the destination, and the duration.
Exclusions – The term significant financial interest does not include:
- Salary or other remuneration from Kalamazoo College
- Income from investment vehicles such as mutual funds or retirement accounts, as long as the investigator does not directly control the investment decisions made in these vehicles
- Income from seminars, lectures, or teaching engagements sponsored by US government agencies, US institutions of higher education, US academic teaching hospitals, US medical centers, or US research institutions affiliated with institutions of higher education
- Income from service on advisory committees or review panels for US government agencies, US institutions of higher education, US academic teaching hospitals, US medical centers, or US research institutes affiliated with institutions of higher education
- Unlicensed intellectual property that does not generate income.
In addition to disclosing any financial conflict of interest at the time of proposal submission, investigators are required to update their financial disclosures during the period of the award, either on an annual basis or as new significant financial interest are obtained.
If the investigator has no significant financial interests to disclose, he or she is required to certify the lack of such interests.
A possible FCOI may not preclude acceptance of the grant or contract in question; but appropriate disclosures or other safeguards may need to be implemented and accepted by both the investigator and the institution. Information received on possible financial interests in a proposed project will be kept strictly confidential, except where public disclosure is a safeguard the investigator and College have agreed upon or such disclosure is required by law or court order.
Guidelines for identifying conflicts of interest. The Director of Faculty Grants will review all financial disclosures, determine whether a conflict of interest exists, and decide what conditions or restrictions (if any) should be imposed to manage, reduce, or eliminate any conflict of interest. A conflict of interest exists when the reviewer reasonably determines that a significant financial interest could directly and significantly affect the design, conduct, or reporting of the proposed or funded research activities.
Examples of conditions or restrictions that might be imposed to manage, reduce or eliminate conflicts of interest include, but are not limited to:
- public disclosure of significant financial interests;
- monitoring of research by independent reviewers;
- modification of the research plan;
- disqualification from participation in the portion of the NSF-funded research that would be
- affected by significant financial interests;
- divestiture of significant financial interests; or
- severance of relationships that create conflicts.
If the Director of Faculty Grants determines that imposing conditions or restrictions would be either ineffective or inequitable, and that the potential negative impacts that may arise from a significant financial interest are outweighed by interests of scientific progress, technology transfer, or the public health and welfare, then the Director of Faculty Grants may allow the research to go forward without imposing such conditions or restrictions.
Compliance. Investigators are expected to comply fully and promptly with the policy.
Reporting. If the Director of Faculty Grants determines that a conflict of interest exists or if the conflict of interest policy is violated, the College will follow federal regulations regarding notification of the sponsoring agency.
Record-keeping. The Director of Faculty Grants will maintain records of all financial disclosures and of all actions taken to resolve conflicts of interest for at least three years beyond the termination or completion of the grant to which they relate, or until the resolution of any PHS or NSF action involving those records, whichever is longer.
Sub-recipients. Kalamazoo College is responsible for ensuring that all sub-recipients comply with federal regulations regarding FCOI. The investigator will notify the Director of Faculty Grants if a subrecipient is affiliated with the project. It is the responsibility of the investigator working with the Director of Faculty Grants to ensure that the sub-recipient provides any necessary FCOI documentation prior to proposal submission. The College shall enter into a written agreement with each sub-recipient that shall specify whether this policy or the applicable policy of the sub-recipient institution will apply to subrecipient investigators. The agreement will specify timing for reporting FCOIs by sub-recipients to Kalamazoo College to enable timely review and reporting of such FCOIs in compliance with funding agency requirements. Kalamazoo College will report any identified FCOI on the part of a sub-recipient to the relevant federal agency within 60 days of disclosure.
NOTE: This policy was originally drafted in 2009 based on PHS regulation 42 CFR Part 50, Subpart F and the NSF Proposal & Award Policies & Procedures Guide, Part II, Award & Administration Guide (AAG) Chapter IV.A and updated according to the NIH 2011 Final Rule in 2012. Parts of this policy were also adapted from the Conflict of Interest Policies at Allegheny College, Hope College, and Smith College, and from Kalamazoo College’s Conflict of Interest Policy for the Board of Trustees and Officers of the College.